We have started the vaccine campaign against the covid and the residents and the professionals of the Residence el Carmen have now had the first dose administered.
The vaccine is guaranteed by the independent regulating agencies which are the ones who authorize the use on the population. The same agéncies regúlate the use of the antibiótics adminístered to children when they are ill, the medicación usted to treat cancer or the anaesthesia adminístered during an operation. No medicine is 100%safe on all the population because each one of us is different. The vaccines are different, most are preventive and are administered on healthy people.
This is why we tolerate less any side effect even is it is insignificant and why this medication is the most regulated, observed and safe that exists.
In conditions of emergency,as in this pandemic,the procedures to authorize the use of new vaccines are accelerated.This is the reason why, in less than a year ,we have a few candidates in the market when the usual process takes more than ten years.
- We have not started from zero,there were some research groups working on some vaccines for similar virus;
- Some of these projects with other coronavirus had already reached the clinical phase I and this previous experience has been kept in mind.
- A great amount of money has been invested ,as has never been done in the history of science ,to carry out these experiments with much more speed;
- There is an international collaboration, also unique in history, among universities, research centres , governments and NGOs;
- The regulating agencies have given priority by means of continuous evaluation, reducing bureaucracy and waiting time but without skipping any stage;
- Clinical phases I and II have been able to overlap and before one was finished the other had already begun;
- Clinical phase III has recruited thousands of volunteers from different countries and different groups, so the results are statiscally more significant than with other trials;
- The production of the vaccines is being carried out assuming a risk.They are being produced for many months without knowing if they will finally be approved,that is the reason why they can appear on the market as soon as authorization is received. Besides,once we know the vaccines are safe and efficient in the previous phases, we must not forget there is another phase IV of observation to continue evaluating its safety and efficiency.
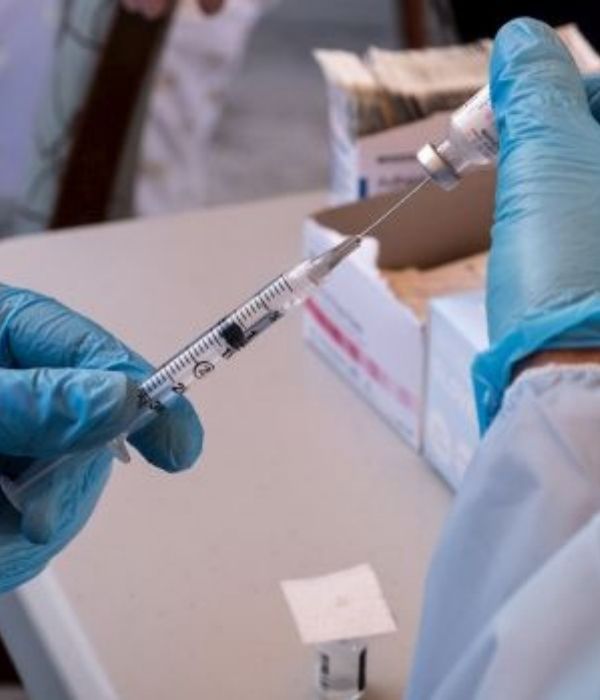
On January 29th 2021 we will receive the second Pfizer dose.In the Residence el Carmen, we will be able to have a Residential Centre adapted to all the guidelines of our Health Centre,Sanitary District and Health Department in favour of caring for our elderly.
At Residencia El Carmen, together with the Residential Unit and the Costa del Sol District, we continue and will continue to ensure the safety and health of our residents; Therefore, on 09/23/2021, the vast majority have received the third booster dose of the Covid-19 vaccine, more gradually the rest of the residents as they enter our residential center and it is mandatory.
According to our agreement,as of today,13/12/2021, all our employees have received the booster shot against the Covid 19.
Next Wednesday,October 5th,2022 we will begin administering the 4th dose of the Covid vaccine and also the flu vaccine to all our residents. This campaign in Andalucía starts with everyone over 80 years of age and also everyone in a nursing home.
Three vaccines will be administered at the same time: the Covid booster,the flu vaccine and the Pneumonia vaccine 13 to anyone who has not been vaccinated previously.
These vaccines are voluntary and enjoy a very high level of recommendation for people who are institutionalized.
ON OCTOBER 9, 2023 WE WILL PROCEED TO VACCINATE THE 5 DOSES OF COVID TO THOSE WHO CORRESPOND TO OUR USERS, PLUS FLU AND PNEUMO 13. This work has been carried out through the district Costa del Sol-Ambulatorio of reference and the direction of the center, where following the guidelines of the Ministry of Health of Andalusia has been bet on focusing it on those over 60 years, as well as vulnerable groups, pregnant women, immunocompromised and health.
ON OCTOBER 9, 2024, THE VACCINATION CAMPAIGN AGAINST INFLUENZA AND COVID-19 WILL START IN ANDALUSIA, for the entire population most vulnerable to these infections and/or their complications, including people institutionalized in social and healthcare centers.
In this campaign, the updated influenza and COVID-19 vaccines will be used, with the currently most circulating variants (JN.1 or KP.2). Following the recommendations of this 2024 campaign, we will take the opportunity to review and update the vaccination against pneumococcus and herpes Zoster if indicated. We confirm in our Residence the total success of this vaccination campaign.



